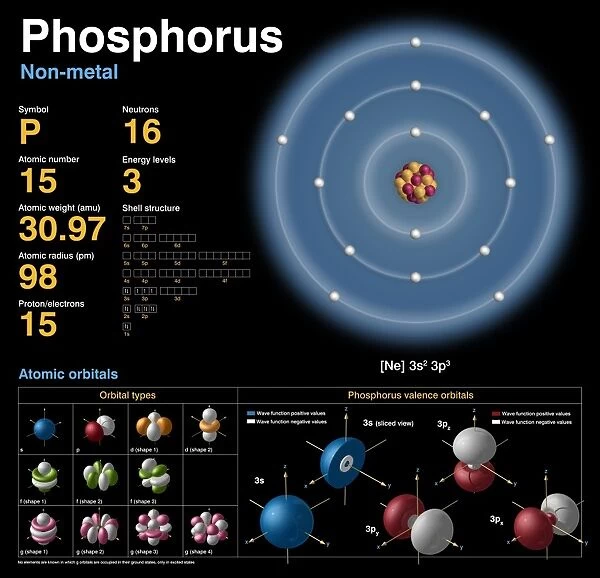
Phosphorus, atomic structure C018 / 3696
![]()

Wall Art and Photo Gifts from Science Photo Library
Phosphorus, atomic structure C018 / 3696
Argon (Ar). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of argon-40 (atomic number: 18), the most common terrestrial isotope of this element. The nucleus consists of 18 protons (red) and 22 neutrons (orange). 18 electrons (white) occupy shells (rings); eight fill the outer (third) electron shell in what is a very stable configuration. The stability of an elements outer (valence) electrons determines its chemical and physical properties. Argon is a noble gas in group 18, period 3, and the p-block of the periodic table. An inert gas formed from the decay of potassium-40, it is used in fluorescent lighting and lasers
Science Photo Library features Science and Medical images including photos and illustrations
Media ID 9262485
© CARLOS CLARIVAN/SCIENCE PHOTO LIBRARY
Atom Atomic Atomic Number Atomic Orbitals Atomic Radius Atomic Weight Chemical Data Chemical Properties Chemical Property Diagram Electron Configuration Electron Number Electron Shell Electronic Electrons Element Elemental Elements Energy Levels Isotope Level Levels Neutron Neutron Number Neutrons Nonmetal Nuclear Nuclear Data Nucleon Nucleons Nucleus Orbital Orbital Types Orbitals Phosphorus Physical Chemistry Physical Properties Physical Property Proton Number Quantum Chemistry Shell Shell Structure Shells Simple Electron Configuration Solid Solids Structural Sub Atomic Subatomic Subshell Subshells Symbol Valence Orbital Wave Function
EDITORS COMMENTS
This print showcases the atomic structure of argon-40, the most abundant isotope of this element found on Earth. The image reveals a detailed diagram of the nuclear composition, electron configuration, and valence orbitals of an argon atom. At its core, 18 protons and 22 neutrons form the nucleus (depicted in red and orange respectively), while 18 electrons occupy various shells or rings around it (shown in white). Notably, eight electrons fill the outermost shell, creating a highly stable configuration. Argon belongs to group 18 and period 3 on the periodic table, making it a noble gas. It falls within the p-block category due to its electronic structure. Interestingly, this inert gas is formed through potassium-40 decay and finds applications in fluorescent lighting and lasers. The visual representation against a black background emphasizes key elements such as symbol illustrations, solid structures, wave functions, atomic weights (in pm), elemental properties like physical chemistry characteristics. Additionally highlighted are subatomic details including neutron numbers and proton numbers along with energy levels associated with different electron shells. Overall, this print provides insight into argon's unique atomic properties that contribute to its chemical behavior as well as physical attributes.
MADE IN THE USA
Safe Shipping with 30 Day Money Back Guarantee
FREE PERSONALISATION*
We are proud to offer a range of customisation features including Personalised Captions, Color Filters and Picture Zoom Tools
SECURE PAYMENTS
We happily accept a wide range of payment options so you can pay for the things you need in the way that is most convenient for you
* Options may vary by product and licensing agreement. Zoomed Pictures can be adjusted in the Cart.